What Happens When Repair Enzymes Do Not Correct Mistakes In Dna?
Dna, similar any other molecule, can undergo a variety of chemical reactions. Because Deoxyribonucleic acid uniquely serves as a permanent copy of the cell genome, however, changes in its structure are of much greater result than are alterations in other cell components, such every bit RNAs or proteins. Mutations tin result from the incorporation of incorrect bases during DNA replication. In addition, various chemical changes occur in DNA either spontaneously (Figure 5.19) or every bit a issue of exposure to chemicals or radiation (Effigy 5.20). Such damage to Deoxyribonucleic acid can block replication or transcription, and tin can result in a high frequency of mutations—consequences that are unacceptable from the standpoint of cell reproduction. To maintain the integrity of their genomes, cells accept therefore had to evolve mechanisms to repair damaged Dna. These mechanisms of Dna repair tin can be divided into two full general classes: (1) direct reversal of the chemical reaction responsible for Deoxyribonucleic acid damage, and (2) removal of the damaged bases followed past their replacement with newly synthesized DNA. Where Deoxyribonucleic acid repair fails, additional mechanisms take evolved to enable cells to cope with the damage.
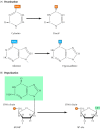
Figure v.19
Spontaneous impairment to Deoxyribonucleic acid. There are ii major forms of spontaneous DNA impairment: (A) deamination of adenine, cytosine, and guanine, and (B) depurination (loss of purine bases) resulting from cleavage of the bond betwixt the purine bases and deoxyribose, (more...)
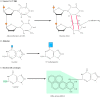
Figure 5.20
Examples of Deoxyribonucleic acid damage induced by radiation and chemicals. (A) UV lite induces the formation of pyrimidine dimers, in which ii adjacent pyrimidines (e.g., thymines) are joined by a cyclobutane ring structure. (B) Alkylation is the improver of methyl (more...)
Direct Reversal of Dna Harm
Most impairment to DNA is repaired past removal of the damaged bases followed past resynthesis of the excised region. Some lesions in DNA, even so, can be repaired by direct reversal of the damage, which may be a more efficient manner of dealing with specific types of Deoxyribonucleic acid harm that occur frequently. Only a few types of DNA harm are repaired in this fashion, particularly pyrimidine dimers resulting from exposure to ultraviolet (UV) calorie-free and alkylated guanine residues that have been modified past the addition of methyl or ethyl groups at the O6 position of the purine band.
UV light is ane of the major sources of harm to DNA and is also the most thoroughly studied form of Dna damage in terms of repair mechanisms. Its importance is illustrated by the fact that exposure to solar UV irradiation is the cause of most all pare cancer in humans. The major type of harm induced past UV low-cal is the formation of pyrimidine dimers, in which side by side pyrimidines on the aforementioned strand of DNA are joined by the germination of a cyclobutane band resulting from saturation of the double bonds between carbons 5 and 6 (see Figure five.20A). The germination of such dimers distorts the structure of the DNA concatenation and blocks transcription or replication by the site of harm, so their repair is closely correlated with the ability of cells to survive UV irradiation. Ane mechanism of repairing UV-induced pyrimidine dimers is direct reversal of the dimerization reaction. The process is called photoreactivation because free energy derived from visible light is utilized to break the cyclobutane ring structure (Effigy 5.21). The original pyrimidine bases remain in DNA, at present restored to their normal land. As might be expected from the fact that solar UV irradiation is a major source of Dna impairment for diverse cell types, the repair of pyrimidine dimers by photoreactivation is common to a variety of prokaryotic and eukaryotic cells, including E. coli, yeasts, and some species of plants and animals. Curiously, yet, photoreactivation is non universal; many species (including humans) lack this mechanism of Deoxyribonucleic acid repair.

Figure v.21
Straight repair of thymine dimers. UV-induced thymine dimers can be repaired by photoreactivation, in which energy from visible low-cal is used to split up the bonds forming the cyclobutane band.
Another course of direct repair deals with damage resulting from the reaction between alkylating agents and Deoxyribonucleic acid. Alkylating agents are reactive compounds that can transfer methyl or ethyl groups to a Deoxyribonucleic acid base, thereby chemically modifying the base (run into Figure 5.20B). A particularly important type of impairment is methylation of the Ohalf-dozen position of guanine, because the product, Ohalf dozen-methylguanine, forms complementary base of operations pairs with thymine instead of cytosine. This lesion tin be repaired past an enzyme (called O6-methylguanine methyltransferase) that transfers the methyl grouping from O6-methylguanine to a cysteine residue in its active site (Figure 5.22). The potentially mutagenic chemical modification is thus removed, and the original guanine is restored. Enzymes that catalyze this direct repair reaction are widespread in both prokaryotes and eukaryotes, including humans.

Figure five.22
Repair of O 6 -methylguanine. O6-methylguanine methyltransferase transfers the methyl group from O6-methylguanine to a cysteine rest in the enzyme'due south active site.
Excision Repair
Although straight repair is an efficient style of dealing with particular types of Deoxyribonucleic acid impairment, excision repair is a more full general means of repairing a wide variety of chemical alterations to DNA. Consequently, the various types of excision repair are the almost important DNA repair mechanisms in both prokaryotic and eukaryotic cells. In excision repair, the damaged DNA is recognized and removed, either as free bases or equally nucleotides. The resulting gap is then filled in past synthesis of a new Deoxyribonucleic acid strand, using the undamaged complementary strand as a template. Three types of excision repair—base-excision repair, nucleotide-excision repair, and mismatch repair—enable cells to cope with a variety of different kinds of Deoxyribonucleic acid damage.
The repair of uracil-containing DNA is a practiced example of base of operations-excision repair, in which single damaged bases are recognized and removed from the DNA molecule (Figure v.23). Uracil tin arise in Dna by two mechanisms: (1) Uracil (equally dUTP [deoxyuridine triphosphate]) is occasionally incorporated in place of thymine during DNA synthesis, and (2) uracil tin can exist formed in DNA by the deamination of cytosine (see Figure v.19A). The second mechanism is of much greater biological significance because it alters the normal pattern of complementary base of operations pairing and thus represents a mutagenic event. The excision of uracil in DNA is catalyzed by Dna glycosylase, an enzyme that cleaves the bond linking the base (uracil) to the deoxyribose of the Dna backbone. This reaction yields gratis uracil and an apyrimidinic site—a sugar with no base attached. Dna glycosylases likewise recognize and remove other abnormal bases, including hypoxanthine formed by the deamination of adenine, pyrimidine dimers, alkylated purines other than O6-alkylguanine, and bases damaged by oxidation or ionizing radiation.

Figure five.23
Base of operations-excision repair. In this example, uracil (U) has been formed by deamination of cytosine (C) and is therefore opposite a guanine (G) in the complementary strand of DNA. The bond between uracil and the deoxyribose is cleaved past a Dna glycosylase, leaving (more than...)
The result of Deoxyribonucleic acid glycosylase action is the formation of an apyridiminic or apurinic site (mostly called an AP site) in DNA. Similar AP sites are formed every bit the outcome of the spontaneous loss of purine bases (see Figure 5.19B), which occurs at a significant charge per unit under normal cellular conditions. For example, each prison cell in the human body is estimated to lose several chiliad purine bases daily. These sites are repaired by AP endonuclease, which cleaves adjacent to the AP site (see Figure 5.23). The remaining deoxyribose moiety is then removed, and the resulting unmarried-base gap is filled by Dna polymerase and ligase.
Whereas DNA glycosylases recognize only specific forms of damaged bases, other excision repair systems recognize a wide diversity of damaged bases that distort the Deoxyribonucleic acid molecule, including UV-induced pyrimidine dimers and bulky groups added to Deoxyribonucleic acid bases as a upshot of the reaction of many carcinogens with DNA (run into Figure 5.20C). This widespread form of DNA repair is known every bit nucleotide-excision repair, because the damaged bases (eastward.m., a thymine dimer) are removed equally part of an oligonucleotide containing the lesion (Effigy v.24).
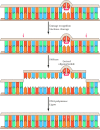
Effigy 5.24
Nucleotide-excision repair of thymine dimers. Damaged DNA is recognized and then broken on both sides of a thymine dimer past iii′ and 5′ nucleases. Unwinding by a helicase results in excision of an oligonucleotide containing the damaged (more than...)
In E. coli, nucleotide-excision repair is catalyzed past the products of three genes (uvrA, B, and C) that were identified considering mutations at these loci issue in farthermost sensitivity to UV light. The protein UvrA recognizes damaged Dna and recruits UvrB and UvrC to the site of the lesion. UvrB and UvrC so carve on the 3′ and 5′ sides of the damaged site, respectively, thus excising an oligonucleotide consisting of 12 or 13 bases. The UvrABC circuitous is oft called an excinuclease, a proper noun that reflects its ability to directly excise an oligonucleotide. The action of a helicase is then required to remove the damage-containing oligonucleotide from the double-stranded DNA molecule, and the resulting gap is filled by DNA polymerase I and sealed by ligase.
Nucleotide-excision repair systems take also been studied extensively in eukaryotes, particularly in yeasts and in humans. In yeasts, as in E. coli, several genes involved in DNA repair (chosen RAD genes for radiation sensitivity) have been identified past the isolation of mutants with increased sensitivity to UV light. In humans, Deoxyribonucleic acid repair genes accept been identified largely by studies of individuals suffering from inherited diseases resulting from deficiencies in the ability to repair Dna damage. The nigh extensively studied of these diseases is xeroderma pigmentosum (XP), a rare genetic disorder that affects approximately one in 250,000 people. Individuals with this disease are extremely sensitive to UV light and develop multiple skin cancers on the regions of their bodies that are exposed to sunlight. In 1968 James Cleaver fabricated the key discovery that cultured cells from XP patients were deficient in the ability to carry out nucleotide-excision repair. This ascertainment not only provided the start link between DNA repair and cancer, only also suggested the employ of XP cells as an experimental arrangement to identify human being DNA repair genes. The identification of human DNA repair genes has been accomplished by studies not only of XP cells, merely likewise of two other human diseases resulting from DNA repair defects (Cockayne's syndrome and trichothiodystrophy) and of UV-sensitive mutants of rodent jail cell lines. The availability of mammalian cells with defects in Dna repair has immune the cloning of repair genes based on the power of wild-type alleles to restore normal UV sensitivity to mutant cells in gene transfer assays, thereby opening the door to experimental assay of nucleotide-excision repair in mammalian cells.
Molecular cloning has now identified seven unlike repair genes (designated XPA through XPG) that are mutated in cases of xeroderma pigmentosum, likewise as in some cases of Cockayne'south syndrome, trichothiodystrophy, and UV-sensitive mutants of rodent cells. Table 5.i lists the enzymes encoded by these genes. Some UV-sensitive rodent cells have mutations in all the same another repair gene, called ERCC1 (for excision repair cross complementing), which has not been found to exist mutated in known human diseases. It is notable that the proteins encoded by these human Deoxyribonucleic acid repair genes are closely related to proteins encoded by yeast RAD genes, indicating that nucleotide-excision repair is highly conserved throughout eukaryotes.
Tabular array 5.1
Enzymes Involved in Nucleotide-Excision Repair.
With cloned yeast and human repair genes bachelor, information technology has been possible to purify their encoded proteins and develop in vitro systems to study the repair process. Although some steps remain to be fully elucidated, these studies have led to the evolution of a basic model for nucleotide-excision repair in eukaryotic cells. In mammalian cells, the XPA protein (and possibly also XPC) initiates repair by recognizing damaged DNA and forming complexes with other proteins involved in the repair procedure. These include the XPB and XPD proteins, which act every bit helicases that unwind the damaged DNA. In addition, the binding of XPA to damaged DNA leads to the recruitment of XPF (as a heterodimer with ERCC1) and XPG to the repair complex. XPF/ERCC1 and XPG are endonucleases, which carve DNA on the 5′ and 3′ sides of the damaged site, respectively. This cleavage excises an oligonucleotide consisting of approximately 30 bases. The resulting gap then appears to be filled in by Deoxyribonucleic acid polymerase δ or ε (in association with RFC and PCNA) and sealed by ligase.
An intriguing feature of nucleotide-excision repair is its relationship to transcription. A connexion between transcription and repair was first suggested past experiments showing that transcribed strands of DNA are repaired more rapidly than nontranscribed strands in both E. coli and mammalian cells. Since DNA damage blocks transcription, this transcription-repair coupling is thought to exist advantageous by allowing the cell to preferentially repair damage to actively expressed genes. In Due east. coli, the mechanism of transcription-repair coupling involves recognition of RNA polymerase stalled at a lesion in the DNA strand being transcribed. The stalled RNA polymerase is recognized by a protein called transcription-repair coupling factor, which displaces RNA polymerase and recruits the UvrABC excinuclease to the site of damage.
Although the molecular machinery of transcription-repair coupling in mammalian cells is not nonetheless known, information technology is noteworthy that the XPB and XPD helicases are components of a multisubunit transcription cistron (called TFIIH) that is required to initiate the transcription of eukaryotic genes (meet Chapter 6). Thus, these helicases appear to be required for the unwinding of DNA during both transcription and nucleotide-excision repair, providing a straight biochemical link between these two processes. Patients suffering from Cockayne'due south syndrome are also characterized from a failure to preferentially repair transcribed Dna strands, suggesting that the proteins encoded by the two genes known to be responsible for this disease (CSA and CSB) part in transcription-coupled repair. In add-on, one of the genes responsible for inherited breast cancer in humans (BRCA1) appears to encode a protein specifically involved in transcription-coupled repair of oxidative DNA damage, suggesting that defects in this type of DNA repair can lead to the development of one of the most common cancers in women.
A 3rd excision repair organisation recognizes mismatched bases that are incorporated during Deoxyribonucleic acid replication. Many such mismatched bases are removed past the proofreading activity of DNA polymerase. The ones that are missed are subject to later correction by the mismatch repair system, which scans newly replicated Dna. If a mismatch is constitute, the enzymes of this repair arrangement are able to identify and excise the mismatched base specifically from the newly replicated Deoxyribonucleic acid strand, assuasive the error to be corrected and the original sequence restored.
In E. coli, the ability of the mismatch repair organisation to distinguish between parental DNA and newly synthesized Deoxyribonucleic acid is based on the fact that Deoxyribonucleic acid of this bacterium is modified by the methylation of adenine residues inside the sequence GATC to form 6-methyladenine (Figure v.25). Since methylation occurs after replication, newly synthesized DNA strands are not methylated and thus tin can be specifically recognized by the mismatch repair enzymes. Mismatch repair is initiated past the protein MutS, which recognizes the mismatch and forms a circuitous with 2 other proteins chosen MutL and MutH. The MutH endonuclease so cleaves the unmethylated Deoxyribonucleic acid strand at a GATC sequence. MutL and MutS and then act together with an exonuclease and a helicase to excise the Deoxyribonucleic acid betwixt the strand break and the mismatch, with the resulting gap being filled by DNA polymerase and ligase.

Effigy five.25
Mismatch repair in E. coli. The mismatch repair organization detects and excises mismatched bases in newly replicated Deoxyribonucleic acid, which is distinguished from the parental strand considering information technology has not yet been methylated. MutS binds to the mismatched base, followed past (more...)
Eukaryotes accept a like mismatch repair system, although the mechanism by which eukaryotic cells identify newly replicated Dna differs from that used by East. coli. In mammalian cells, information technology appears that the strand-specificity of mismatch repair is determined past the presence of unmarried-strand breaks (which would be nowadays in newly replicated DNA) in the strand to exist repaired (Figure 5.26). The eukaryotic homologs of MutS and MutL and then bind to the mismatched base and direct excision of the DNA between the strand pause and the mismatch, every bit in East. coli. The importance of this repair organisation is dramatically illustrated by the fact that mutations in the man homologs of MutS and MutL are responsible for a common type of inherited colon cancer (hereditary nonpolyposis colorectal cancer, or HNPCC). HNPCC is 1 of the most common inherited diseases; it affects as many as one in 200 people and is responsible for about fifteen% of all colorectal cancers in this country. The human relationship betwixt HNPCC and defects in mismatch repair was discovered in 1993, when two groups of researchers cloned the human being homolog of MutS and establish that mutations in this factor were responsible for about one-half of all HNPCC cases. Subsequent studies have shown that most of the remaining cases of HNPCC are caused by mutations in one of iii human genes that are homologs of MutL.

Figure 5.26
Mismatch repair in mammalian cells. Mismatch repair in mammalian cells is similar to E. coli, except that the newly replicated strand is distinguished from the parental strand because it contains strand breaks. MutS and MutL demark to the mismatched base of operations (more than...)
Postreplication Repair
The direct reversal and excision repair systems deed to right DNA damage before replication, so that replicative DNA synthesis tin proceed using an undamaged Dna strand as a template. Should these systems fail, even so, the prison cell has alternative mechanisms for dealing with damaged Dna at the replication fork. Pyrimidine dimers and many other types of lesions cannot exist copied past the normal action of DNA polymerases, and so replication is blocked at the sites of such damage. Downstream of the damaged site, however, replication can be initiated again by the synthesis of an Okazaki fragment and can proceed along the damaged template strand (Figure 5.27). The result is a daughter strand that has a gap opposite the site of harm to the parental strand. I of two types of mechanisms may be used to repair such gaps in newly synthesized DNA: recombinational repair or fault-decumbent repair.

Effigy 5.27
Postreplication repair. The presence of a thymine dimer blocks replication, but DNA polymerase can featherbed the lesion and reinitiate replication at a new site downstream of the dimer. The event is a gap opposite the dimer in the newly synthesized Deoxyribonucleic acid (more than...)
Recombinational repair depends on the fact that one strand of the parental DNA was undamaged and therefore was copied during replication to yield a normal daughter molecule (see Figure 5.27). The undamaged parental strand can be used to fill up the gap opposite the site of damage in the other girl molecule by recombination between homologous DNA sequences (come across the next section). Because the resulting gap in the previously intact parental strand is reverse an undamaged strand, information technology can be filled in past Dna polymerase. Although the other parent molecule still retains the original damage (east.yard., a pyrimidine dimer), the damage now lies opposite a normal strand and can be dealt with later by excision repair. By a similar mechanism, recombination with an intact DNA molecule tin can be used to repair double strand breaks, which are oftentimes introduced into DNA by radiations and other damaging agents.
In error-prone repair, a gap opposite a site of DNA damage is filled by newly synthesized Dna. Since the new Dna is synthesized from a damaged template strand, this form of DNA synthesis is very inaccurate and leads to frequent mutations. Information technology is used just in bacteria that have been subjected to potentially lethal conditions, such equally all-encompassing UV irradiation. Such treatments induce the SOS response, which may exist viewed equally a mechanism for dealing with extreme ecology stress. The SOS response includes inhibition of cell sectionalisation and induction of repair systems to cope with a high level of Deoxyribonucleic acid damage. Under these conditions, error-prone repair mechanisms are used, presumably as a way of dealing with damage so extensive that cell death is the only alternative.
![]()
Box
Molecular Medicine : Colon Cancer and Deoxyribonucleic acid Repair.
What Happens When Repair Enzymes Do Not Correct Mistakes In Dna?,
Source: https://www.ncbi.nlm.nih.gov/books/NBK9900/
Posted by: waterswheines.blogspot.com


0 Response to "What Happens When Repair Enzymes Do Not Correct Mistakes In Dna?"
Post a Comment